Axon Guidance and Neurogenetics
Scientists involved: Lukas Geschwender, Lukas Lachenmaier, Sebastiana Ruisi, Prof. Dr. Martin Bastmeyer, Dr. Franco Weth
Funding: Landesgraduiertenförderung, Karlsruhe School of Optics and Photonics (KSOP)
Like in artificial neuronal networks, all sensory, cognitive and motor capabilities of the brain rely on its connectivity matrix, which is of bewildering complexity. In contrast to its artificial counterpart, however, substantial parts of the circuitry of this marvelous computing network arise by directed hard-wiring during embryonic development. Outgrowing axons are guided to their appropriate targets by genetically encoded signals, which are read by a growth cone at the tip of the growing axon. The molecular mechanisms of growth cone navigation have remained poorly understood. In our group, we try to contribute to the elucidation of the mechanistic underpinnings of these processes. Various human neurodevelopmental disorders are thought to involve mis-wiring of brain circuits. Therefore, in a parallel, translational approach, we also address genetic models of human neurodevelopmental disorders using schizophrenia as an example
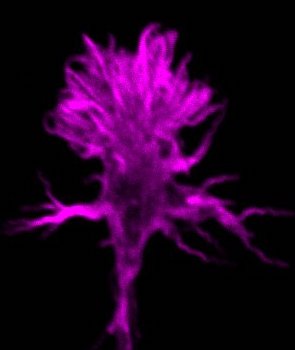 |
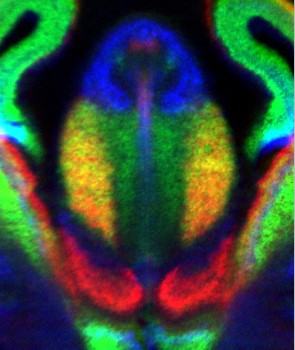 |
| Axon Guidance | Neurogenetics |